Chronic myeloid leukemia (CML) is caused by the BCR-ABL1 tyrosine kinase, the product of the Philadelphia chromosome. In the chronic phase of CML (CP-CML), myeloid cells expand, but maintain differentiation. BCR-ABL1 tyrosine kinase inhibitors (TKIs) frequently induce deep responses, but do not eliminate primitive, quiescent CML stem cells (LSCs), whose survival is independent of BCR-ABL1 kinase activity.
As a result, most patients require lifelong TKI therapy to prevent recurrence of active CML. In ~10% of patients, a block of differentiation converts CP-CML to blast phase CML (BP-CML), an acute leukemia of typically myeloid phenotype. Blasts from many BP-CML cases have acquired BCR-ABL1 independence and are resistant to TKIs.
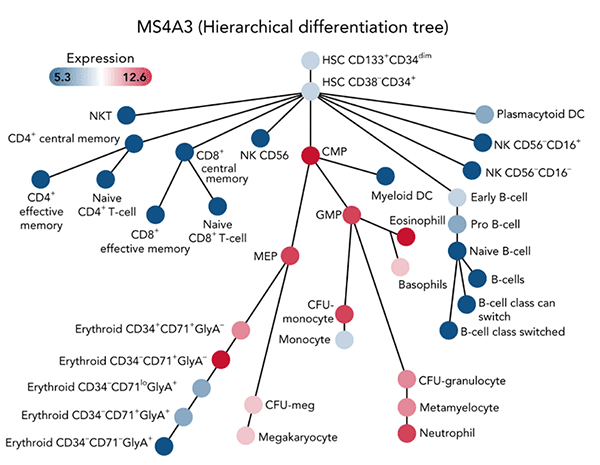 MS4A3 expression on a hierarchical differentiation tree, based on BloodSpot illustration of the dataset GSE24759.
MS4A3 expression on a hierarchical differentiation tree, based on BloodSpot illustration of the dataset GSE24759.Our premise is that a differentiation block is central to the BCR-ABL1 independence that characterizes the extremes of the clinical CML spectrum: residual leukemia in TKI responders and TKI-resistant BP-CML.
To identify effector molecules mediating BCR-ABL1 independence and differentiation block, we performed a meta-analysis of published CML transcriptomes. We discovered that expression of MS4A3, a member of the MS4A (membrane-spanning four A) family of signaling proteins, is profoundly reduced in CML quiescence, TKI resistance and BP-CML. MS4A3’s role in normal and leukemic hematopoiesis is largely unknown. Previous reports implicated MS4A3 as a cell cycle inhibitor in U937 leukemia cells, and an early marker of myelomonocytic differentiation.
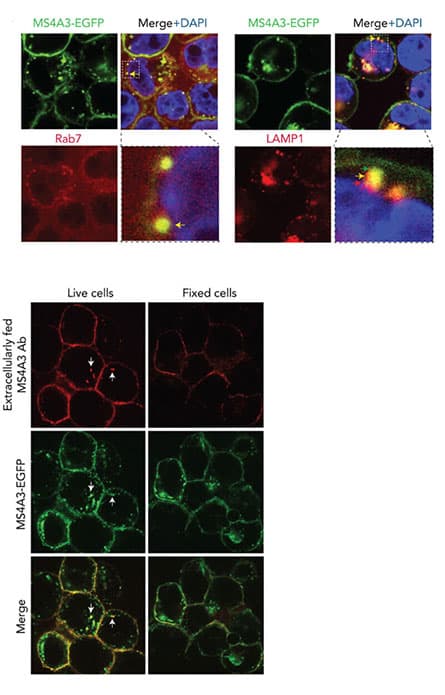 MS4A3 traffics between plasma membrane and endosomes to regulate the endocytosis surface-bound protein.
MS4A3 traffics between plasma membrane and endosomes to regulate the endocytosis surface-bound protein.Our preliminary data show that MS4A3 knockdown inhibits differentiation, enhances TKI resistance and promotes survival of CML stem and progenitor cells (LSPCs) in vitro and in vivo. By contrast, ectopic MS4A3 expression increases differentiation and TKI sensitivity. Consistent with these data, low MS4A3 expression correlates with shorter survival in CP-CML. At a mechanistic level, MS4A3 enhances IL-3 and GM-CSF signaling in CML CD34+ cells by promoting endocytosis of β common chain (βc) cytokine receptors, suggesting that MS4A3 is not merely a marker, but a positive regulator of myeloid differentiation.
In support of this, delivery of MS4A3-containing nanoparticles to BP-CML CD34+ cells induce differentiation and reduces colony formation. These data suggest that MS4A3 promotes differentiation by enhancing βc receptor endocytosis and IL-3/GM-CSF signaling. TKI resistant LSPCs reduce MS4A3 to blunt response to differentiation-inducing cytokines to maintain an undifferentiated, therapy-resistant state, possibly coopting a physiological stress response mechanism.
The heat map and diagram representing CpG methylation levels in the MS4A3 promoter region, as detected by DNA bisulfate conversion and patch PCR sequencing on DNA of CD34+ cells from CB, adult BM, CP-CML and BP-CML samples.
We will work to: (1) Delineate how MS4A3 regulates endocytosis and signaling of βc cytokine receptors, (2) Determine the function of MS4A3 in normal hematopoiesis, and (3) Delineate the role of MS4A3 in CML hematopoiesis and as a therapeutic agent in CML.