
Cellular & Biotherapies


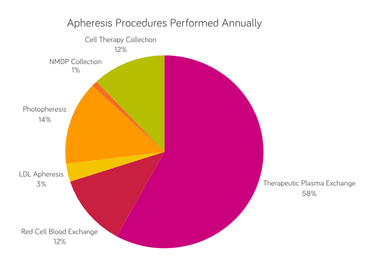
During plasma exchange therapy, a patient’s plasma is removed and replaced with 5% albumin and/or donated plasma. TPE is used for a wide range of indications, including antibody-mediated syndromes in neurology, nephrology, hematology and organ transplantation.
- Available in Wisconsin and Michigan
A procedure in which abnormal red blood cells (RBCs) are removed and replaced with donated RBCs. RBCX is performed for acute episodes and chronic management in sickle cell disease and in the case of certain infections (e.g., malaria and babesiosis) in which RBCs are damaged.
- Available in Wisconsin and Michigan
During this procedure, an excessive amount of white blood cells is rapidly removed in disease causing hyperleukocytosis, such as leukemia, which may cause complications such as stroke, respiratory failure and/or renal failure.
- Available in Wisconsin and Michigan
A procedure in which an excessive amount of platelets is rapidly removed in diseases like essential thrombocytosis, which may cause complications, such as stroke or heart attack.
- Available in Wisconsin and Michigan
A procedure in which white blood cells are separated from a patient’s blood and treated with a medication called Methoxsalen, which makes them sensitive to ultraviolet light. The treated white blood cells are exposed to ultraviolet light and returned to the patient. Photopheresis may be used to treat graft vs. host disease, cutaneous T-cell lymphoma and organ transplant rejection.
- Available in Wisconsin and Michigan
A procedure in which LDL cholesterol is rapidly removed for chronic treatment of familial hypercholesterolemia. LDL apheresis can also be used to treat focal segmental glomerulosclerosis (FSGS) when it affects the native kidney or after a kidney transplant.
- Available in Wisconsin only

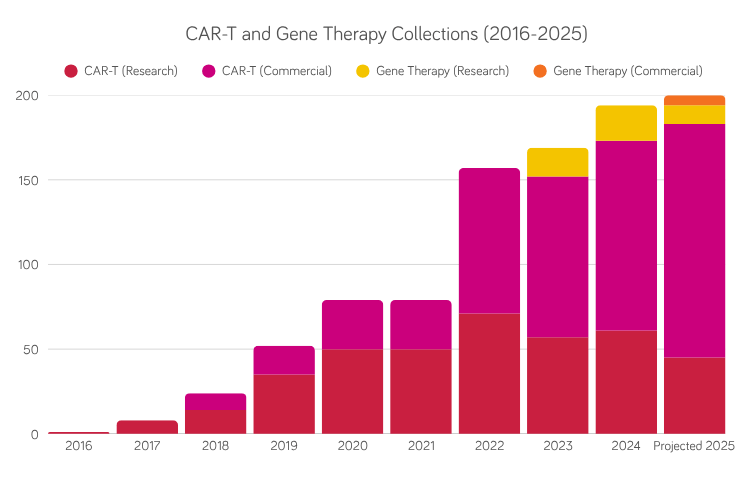
A procedure in which MNCs are collected from a donor as the source material for development of individualized cell therapy products.
- Agreement for Freezing and Storage of Cellular Therapy Products
- BMT Infusion Request
- Declaration of Urgent Medical Need
- Infectious Disease Marker (IDM) Testing
- Order to Release Cryopreserved Products
- Post Collection Amendment of Eligibility
- Cellular Therapy Product Processing Request
- Unpacking and Storing CBU at Hospital


